Theme: Advancing role of microbes
Microbiome R&D 2018
- Mayor Invitation
- Welcome Message
- Microbiome R&D 2018
- Sessions/Tracks
- About Microbiome
- Travel Guide
- Market Analysis

Dear Speakers, Delegates, Exhibitors & Sponsors,
Conference Series LLC Ltd welcomes you to join the International Conference on Microbiome R&D and Biostimulants (Microbiome R&D 2018) during October 15-16, 2018. The conference will be held in the city of Ottawa, Canada
The theme of Microbiome R&D 2018 is to “Advancing role of microbes”
The Microbiome R&D 2018 Conference is an International conference of 2 days, providing the most up-to-date information, research, guidance, and policy discussion for professionals in the Microbiome and Biostimulants field. It is your opportunity to learn and network with friends and colleagues from around the globe while having fun at the same time.
We very much look forward to seeing you in Ottawa, Canada on October 15-16.
In case you have a question about the conference, contact Ms. Laura Smith, the conference manager of this conference at biologicalmicrobiome@protonmail.com or you can call at +1-800-014-8923.
Regards,
Conference Committee
Microbiome R&D 2018
Details of Microbiome R&D 2018 Conference in Canada:
Conference series LLC Ltd is organizing Microbiome R&D 2018 Conference in 2018 at Ottawa, Canada. We organize Microbiome Meetings in the fields related to human microbiota, gut microbiota, prebiotics, probiotics, pharmacogenomics, cancer development, autoimmune disorders, skin microbiome, oral microbiome, pediatric microbiome, soil microbiome, biostimulants, hydroponics, beneficial microbes.
|
Conference Name |
Place |
Date |
|
Microbiome R&D 2018 |
Ottawa, Ontario, Canada |
October 15-16, 2018 |
International Conference on Microbiome R&D and Biostimulants are scheduled to be held during October 15-16, 2018 in Ottawa, Canada. This Microbiome R&D 2018 anticipates more than 200 participants around the globe with thought-provoking Keynote lectures, Oral talks and Poster presentations, Symposia, Workshops, Exhibitions and Career development programs. The attending delegates include Editorial Board Members of related ConferenceSeries Journals. This is an excellent opportunity for the delegates from Universities and Institutes to interact with the world class Scientists. The intending participants can confirm their participation by registering at Microbiome R&D 2018 along with your colleagues.
Microbiome R&D 2018 is one of the well-established conferences among Microbiology Conferences organized by Conference series LLC Ltd
Conference series LLC Ltd Organizes 3000+ Global Events Every Year across the USA, Europe & Asia with support from 1000 more scientific societies and Publishes 700+ Open access journals which contain over 100000 eminent personalities, reputed scientists as editorial board and organizing committee members. The conference series LLC Ltd website will provide you list and details about the conferences organize worldwide.
Why to Attend???
Microbiome R&D 2018 Conference is a multidisciplinary program with broad participation with members from around the globe focused on learning about microbiology research and its advances. This is your best opportunity to reach the largest assemblage of participants from microbiology community that is from academia, microbiology entities, medical groups, labs, related associations, societies and also from government agencies, pharmaceutical, biomedical and medical device industries. This conference conduct presentations, distribute information, meet with current and potential scientists, make a splash with new clinical research developments, and receive name recognition at this 2-days event. World-renowned speakers and the most recent techniques, developments, the newest updates in Microbiome R&D are hallmarks of this conference.
Who Should Attend and Who You’ll Meet
Directors/Senior Directors/Executive Directors and Vice Presidents/Senior Vice Presidents/Executive Vice Presidents and Heads/Leaders/Partners of
- Microbiology Research Sites
- Pathologists and Immunologists
- Research Scientists
- Pharma/Biotech and Medical Device industries
- Hospitals, Labs & Associations
- Horticulture specialists
- Arborists
- Farm technicians
- Hydroponics Growers
- Environmental specialists
Medical Directors, Principal Investigators, Methodologists, and other clinical research professionals along with Academicians: University Faculties like Directors, Senior Professors/Assistant Professors/ Associate Professor, Research Scholars, investors, scientists who are related to Microbiome R&D. Pathologists, Doctors, Director and Managers from business organizations.
Benefits of attending Microbiome R&D Conference?
· An opportunity to meet the mentors across the world face-to-face.
· Sharing ideas, challenges and plan something interesting for future
· B2B Meetings.
· To meet the experts in the field of Microbiome R&D.
· To share the knowledge with doctors and Scientists.
· To gain advanced knowledge in Microbiome R&D.
· To meet investors from a different sector of Healthcare.
· To develop collaborations between Academic and Business.
· Thought to provoke talks by Doctors, Professors, Ph.D.'s and Young Researchers.
Microbiome R&D 2018 ardently encourages patrons, benefactors, pioneers, and exhibitors from across the world to convene you all to attend and register for the “International Conference on Microbiome R&D and Biostimulants (Microbiome R&D 2018) which is going to be venerated from October 15-16, 2018 at Ottawa, Canada.
The organizing committee is endowing for a rather stimulating and illuminative conference programs subsuming Plenary Lectures, Symposia, Workshops on a myriad of topics, Poster Presentations and an assortment of programs for participants from over the globe. We invite you to accompany us at the Microbiome R&D 2018, where you are assured to have a substantially worthwhile experience with ecumenical scholars.
The Dynamic Microbiome:
Our genomes are essentially static, our microbiomes are inherently dynamic. The microbial communities we harbor in our bodies change throughout our lives due to many factors, including maturation during childhood, alterations in our diets, travel, illnesses, and medical treatments. Moreover, there is mounting evidence that our microbiomes change us, by promoting health through their beneficial actions or by increasing our susceptibility to diseases through a process termed dysbiosis. microbiomes change us, by promoting health through their beneficial actions or by increasing our susceptibility to diseases through a process termed dysbiosis Recent technological advances are enabling unprecedentedly detailed studies of the dynamics of the microbiota in animal models and human populations. Dramatic changes can occur with diseases, such as infections or inflammatory bowel disease . Medical interventions, such as antibiotic treatments can also profoundly affect the microbiota. Longitudinal microbiome studies are beginning to yield exciting insights into the dynamic behaviors of the microbiota, including microbial succession events during infant gut maturation, normal temporal variability in healthy adults, responses over time to perturbations such as antibiotics and dietary changes, and dysbiotic alterations that presage symptomatic disease. Computational tools for analyzing microbiome timeâ€series data are another area where we're likely to see tremendous growth. we are already beginning to see more sophisticated techniques being applied to analyzing microbiome timeâ€series data, such as nonâ€parametric Bayesian models and dynamical systems theory, interâ€individual microbiome variability in humans, which can obscure any common signal present. Computational techniques that could model interâ€individual variability while automatically capturing commonalities at appropriate levels in ecosystems would be very powerful. The dynamic microbial battles and alliances that are being played out in our bodies provide a fascinating counterâ€point to our rather staid, stable genomes.
Microbiome as a human organ:
Microbiome: Deciphering the Last Organ of the Human Body
The human organism is a complex structure composed of cells belonging to all three domains of life on Earth, Eukarya, Bacteria and Archaea, as well as their viruses. Bacterial cells of more than a thousand taxonomic units are condensed in a particular functional collective domain, the intestinal microbiome. The microbiome constitutes the last human organ under active research microbiome is readily inherited. Like any other organ, the microbiome has physiology and pathology, and the individual health might be damaged when its collective population structure is altered. The diagnostic of microbiomic diseases involves metagenomic studies. The therapeutics of microbiomeâ€induced pathology include microbiota transplantation, a technique increasingly available microbiome can be regarded as a human organ from the physiological standpoint Perhaps we can envisage ‘microbiomology’ as a future specialty. Devoted to the study of the physiology, pathology, diagnostics, therapy and prevention of alterations of the community structure of the microbiome. The importance of the microbiome has been highlighted by the microbial ‘abnormalities’ found in pathological conditions such as inflammatory bowel diseases, obesity or malnutrition. Diagnosis of microbiome diseases is based at present on full metagenomic DNA sequencing and computational advances that can inform about and differentiate core microbiota and changing microbiota These ‘diagnostic’ techniques should also be able to evaluate the role of mobile genetic elements, which deeply influence the connectivity of the microbiome. The therapy of microbiome diseases will be part of future interventions based on ecoâ€evo drugs and strategies. Addressing microbiome restoration by transplantation is crucial to advance in the curing of microbiome diseases A more advanced field of research in the therapy of microbiome diseases will be the discovery of drugs acting on host–microbiome and intraâ€microbiome signals and interactions.
Microbiota in early life shapes the immune system:
Microbial colonization of mucosal tissues during infancy plays an instrumental role in the development and education of the host mammalian immune system. These early-life events can have long-standing consequences: facilitating tolerance to environmental exposures or contributing to the development of disease in later life, including inflammatory bowel disease, allergy, and asthma. Recent studies have begun to define a critical period during early development in which disruption of optimal host-commensal interactions can lead to persistent and in some cases irreversible defects in the development and training of specific immune subsets.
The Microbiome as a Therapeutic Target for Metabolic Diseases:
Large-scale alterations of the gut microbiota and its microbiome (gene content) are associated with Metabolic diseases, especially type 2 diabetes and obesity and are responsive to weight loss, are growing global healthâ€care concerns. Gut microbes can impact host metabolism via signaling pathways in the gut, with effects on inflammation, insulin resistance, and deposition of energy in fat stores. New therapeutic approaches are urgently needed to address the emerging epidemic of obesity and diabetes. Restoration of the gut microbiota to a healthy state may ameliorate the conditions associated with obesity and help maintain a healthy weight. The microbiome, the collective genomic and metabolic potential of the gut microbiota, may have a key role in many chronic diseases through its mitigation of immuneâ€inflammatory responses.
Pharmacogenomics:
Pharmacogenomics can play an important role in identifying responders and non-responders to medications, avoiding adverse events, and optimizing drug dose. Genetic polymorphisms in drug-metabolizing enzymes, transporters, receptors, and other drug targets have been linked to interindividual differences in the efficacy and toxicity of many medications. Pharmacogenomic studies are rapidly elucidating the inherited nature of these differences in drug disposition and effects, thereby enhancing drug discovery and providing a stronger scientific basis for optimizing drug therapy on the basis of each patient's genetic constitution.
The role of the microbiome in cancer development and therapy:
The human body harbors enormous numbers of microbiota that influence cancer susceptibility, in part through their prodigious metabolic capacity and their profound influence on immune cell function. Even larger numbers of malignancies are associated with an altered composition of commensal microbiota (dysbiosis) based on microbiome studies using metagenomic sequencing. Microbiota can alter cancer susceptibility and progression by diverse mechanisms, such as modulating inflammation, inducing DNA damage, and producing metabolites involved in oncogenesis or tumor suppression. Microbiota can be manipulated for improving cancer treatment. By incorporating probiotics as adjuvants for checkpoint immunotherapy or by designing small molecules that target microbial enzymes, microbiota can be harnessed to improve cancer care. The efficacy of chemotherapy/immunotherapy likely depends on an individual's microbiota, maintenance of microbial diversity is critical for human health. Steps should be taken to prevent indiscriminate antibiotic usage. Furthermore, encouraging a diverse, plantâ€based diet facilitates microbial diversity.
Interplay between diet, gut microbiota, epigenetic events, and colorectal cancer:
Diet and energy balance influence CRC by multiple mechanisms. They modulate the composition and function of gut microbiota, which have a prodigious metabolic capacity and can produce oncometabolites or tumorâ€suppressive metabolites depending, in part, on which dietary factors and digestive components are present in the GI tract. Gut microbiota also have a profound effect on immune cells in the lamina propria, which influences inflammation and subsequently CRC. The nutrient availability, which is an outcome of diet and energy balance, determines the abundance of certain energy metabolites that are essential coâ€factors for epigenetic enzymes and therefore impinges upon the epigenetic regulation of gene expression. Aberrant epigenetic marks accumulate during CRC and epimutations that are selected for drive tumorigenesis by causing transcriptome profiles to diverge from the cell of origin. In some instances, the above mechanisms are intertwined as exemplified by dietary fiber being metabolized by colonic bacteria into butyrate, which is both a shortâ€chain fatty acid (SCFA) and a histone deacetylase (HDAC) inhibitor that epigenetically upregulates tumorâ€suppressor genes in CRC cells and antiâ€inflammatory genes in immune cells.
Pediatric Microbiome [Vaginal Delivery Versus C-Section]:
There are two ways by which a baby can come into this world: vaginally or by Cesarean delivery. The vast majority of our critical gut microbiome bacteria from our mothers during birth and breastfeeding. The method of delivery impacts the baby’s microbiome, with vaginal delivery (VD) having a strong, beneficial effect and cesarean delivery (CD) reducing the number and diversity of beneficial bacteria. Cesarean delivery poses a health risk for newborns by way of changes in the gut microbiota there microbial species or genera that are uniformly present in all vaginally delivered infants and uniformly absent in all Cesareanâ€born babies prolonged effects of birth mode on microbiota composition that coâ€occurred with Cesarean delivery the mother’s collective microbiome is healthy and that baby receives as many essential bacteria as possible from her during birth and breastfeeding, as her microbiome forms the foundation for baby’s microbiome, which is essential for health, development, and metabolism from infancy through childhood. The infant’s gut microbiome is of critical importance since its bacteria build and strengthen baby’s immune system. the microbiomes of babies born via vaginal delivery (VD) and via cesarean delivery (CD) are different, due to the different microbiomes they are receiving from their mother and their physical environment, during a vaginal birth, the baby receives maternal vaginal, intestinal and fecal bacteria present in the birth canal; these bacteria are augmented with bacteria from mother’s skin, oral and breast milk microbiomes through holding, kissing and breastfeeding. A baby born by CD does not receive these initial bacteria, but rather the bacteria present in his mother’s skin and the hospital environment which contains harmful bacteria subsequently, CD babies have significantly lower bacterial amounts and diversity than vaginally born babies, which is less than ideal. Since they were not seeded with the optimal array of bacteria at birth, their immune systems may fail to develop properly, leaving them more susceptible to pathogens. Scientists speculate this may account for the increased long-term risk and incidence of chronic, non-communicable diseases (including allergies, asthma, obesity and autoimmune diseases) among babies born via cesarean section.
Role of microbial communities in the pathogenesis of periodontal diseases and caries (Oral Microbiome):
There is an increasing evidence to suggest that both caries and periodontal disease represent dysbiotic states of the oral microbiome, The human oral microbiome currently comprises 600–700 taxa, but estimates suggest that overall species numbers may turn out to be higher. Within the oral cavity, groups of microbial species become arranged into surfaceâ€localized communities that vary considerably in the the composition according to sites of the the establishment. Factors such as nutrient availability, pH, toxic metabolites, shear forces and host conditions contribute to modeling the structure and activities of these oral microbial communities. A repertoire of stable dysbiotic states may occur in both caries and periodontitis involving different microbial community structures with potentially similar functional properties. The mode of acquisition of oral microbial communities may be less passive than previously recognized but once established remains relatively stable within an individual although there are very significant site variations, a number of issues pertinent to the community organization and functional activity of the bacterial populations resident on supra†and subgingival tooth surface and the influence of these populations on disease.
The Skin microbiome:
The Nobel laureate Joshua Lederberg suggested using the term ‘human microbiome’ to describe the collective genome of our indigenous microorganisms (microflora) colonizing the whole body. The skin is a complex barrier organ made of a symbiotic relationship between microbial communities and host tissue via complex signals provided by the innate and the adaptive immune systems. It is constantly exposed to various endogenous and exogenous factors which impact this balanced system potentially leading to inflammatory skin conditions comprising infections, allergies or autoimmune diseases started using modern methods such as pyrosequencing assays of bacterial 16S rRNA genes to identify and characterize the different microorganisms present on the skin, to evaluate the bacterial diversity and their relative abundance and to understand how microbial diversity may contribute to skin health and dermatological conditions, Three main sampling methods are currently used to harvest the resident skin microbiota The skin barrier and the microbiota act like a shield that protects the body against external aggressions. There is a balanced interplay between the host and resident and/or transient bacterial populations. This balance is continuously affected by intrinsic (host) and extrinsic (environmental) factors that alter the composition of skin microorganism communities and the host skin barrier function. Altering this equilibrium is called dysbiosis. Underlying pathobiology or genetically determined variations in stratum corneum properties might result in a dysbiosis that changes the abundance and diversity of commensal species, which disturbs skin barrier function and aggravates chronic skin diseases such as atopic dermatitis and psoriasis dysbiosis does not only occur between bacteria, disequilibrium between bacteria and commensal fungi strains on the scalp has been observed in subjects prone to dandruff the impact of environmental factors such as climate, including temperature and UV exposure but also of lifestyle, including alcoholism or nutrition on microbial communities remains to be elucidated. Indeed, ultraviolet B and C light have been reported to be bactericidal, while excessive alcohol consumption has been shown to diminish host resistance and nutrient and vitamin deficiency has been shown to impact on the skin microbiota balance, resulting in infection and skin barrier disturbance, Cosmetics, hygiene products, makeup, and moisturizers have also been implicated in modifying the skin microbiome, Radiotherapy and chemotherapy used to treat cancer may also impact the microbiota. Therefore, improving the knowledge about the skin microbiome may open new perspectives in the management of the healthy and diseased skin and of its microbiome.
Skincare products with probiotics worth the hype:
Skin is subjected to harsh environmental conditions that favor the growth of primarily gram-positive organisms, resident bacteria are capable of reproducing and are commensal with the host when skin is healthy. When skin is compromised, resident bacteria can become pathogens as is seen in acne and folliculitis. The skin microbiome is influenced by pH, sebum content, barrier function, and hydration, alterations in skin microflora play a significant role in conditions such as atopic dermatitis, psoriasis, acne and skin cancer.
Probiotics are live bacterial cultures that, when applied topically, influence the composition of skin microflora. Acidifying the skin discourages the growth of most pathogens favoring the the growth of resident flora. Probiotic strains produce potent antimicrobials such as bacteriocidins, organic acids, and, H2O2 that prevent pathogen adhesion. Lactic acid acts as a natural moisturizing factor and antimicrobial and acts on epidermal and dermal remodeling
Prebiotics are non-digestible plant-based carbohydrates that discourage the growth of pathogens while preserving beneficial bacteria. Prebiotics can be readily incorporated into skin care products and are an excellent alternative to live bacteria. Bacterial cell lysates are also used in cosmetic formulations.
Gut Microbiota :
Trillions of microbes inhabit the human intestine, forming a complex ecological community that influences normal physiology and susceptibility to disease through its collective metabolic activities and host interactions. Understanding the factors that underlie changes in the composition and function of the gut microbiota will aid in the design of therapies that target it. This goal is formidable. The gut microbiota is immensely diverse, varies between individuals and can fluctuate over time — especially during disease and early development. Viewing the microbiota from an ecological perspective could provide insight into how to promote health by targeting this microbial community in clinical treatments.
The gut metagenome is the aggregate of all the genomes of gut microbiota. The gut microbiota plays a key role in digestion, metabolism and immune function, and has a widespread impact beyond the gastrointestinal tract. Changes in the biodiversity of the gut microbiota are associated with far-reaching consequences on host health and development. Diet, functional foods, and gut microbiota transplantation are areas that have yielded some therapeutic success in modulating the gut microbiota and warrant further investigation of their effects on various disease states.
Human Microbiota :
The collection of microbes living in and on our body - have a significant impact on human health and well-being. They have been associated with numerous diseases, yet we have barely understood their role in the context of lifestyle and genetics. Various initiatives are underway around the world to survey the human microbiota at several body sites, characterize them, understand their interactions with the human hosts, elucidate their role in diseases, and design possible therapeutic or dietary interventions.
Modulation of Microbiota :
Microbiota modulation appears as an interesting tool in the prevention and/or treatment of the dysbiosis associated with obesity and metabolic disorders. Colonization of mucosal surfaces is characterized by fluctuating changes in microbial diversity during the first few years of life, until reaching a point of equilibrium that remains relatively stable throughout adulthood in the absence of environmental insults, immune maturation is likely influenced directly and/or indirectly by the presence of commensal microbes the early-life ecological succession of mucosal colonization occurs concomitantly with the development, expansion, and education of the mucosal immune system.
Prebiotics and Probiotics:
Prebiotics and probiotics may be useful in achieving positive effects which include the enhanced immune function, improved colonic integrity, decreased inci dence and duration of intestinal infections, down-regulated allergic response, improved digestion, and elimination. Probiotics and prebiotics share a unique role in human nutrition, largely centering on manipulation of populations or activities of the bacteria that colonize our bodies. There is a need to consolidate the basic and applied research on probiotics and prebiotics into useful tools for food and nutrition, for example, LAB are gram-positive nonpathogenic bacteria that are widely distributed in the nature have long been used in food processing the LcS is widely used in the production of probiotic dairy products and is also used as a food ingredient. The probiotic bacteria have the potential to augment or modify the host immune function through the regulation of host immune cells.
Bioinformatics for Microbiome :
Microbes inhabit virtually all sites of the human body, yet we know very little about the role they play in our health. In recent years, there has been increasing interest in studying human-associated microbial communities, particularly since microbial dysbioses have now been implicated in a number of human diseases. Recent advances in sequencing technologies have made it feasible to perform large-scale studies of microbial communities, providing the tools. Rapidly developing sequencing methods and analytical techniques, the human microbiome on different spatial and temporal scales, including daily time series datasets spanning months. Furthermore, emerging concepts related to defining operational taxonomic units, diversity indices, core versus transient microbiomes, are enhancing our ability to understand the human microbiome.
16S sequencing & Analysis :
16S ribosomal RNA (or 16S rRNA) is the component of the 30S small subunit of a prokaryotic ribosome that binds to the Shine-Dalgarno sequence. 16S rRNA sequencing has been used to characterize the complexity of microbial communities at each body sites, and to determine whether there is a core microbiome. The 16S rRNA sequence contains both highly conserved and variable regions. These variable regions, nine in number (V1 through V9), are routinely used to classify organisms according to phylogeny, making 16S rRNA sequencing particularly useful in metagenomics to help identify taxonomic groups present in a sample.
Animals in a bacterial world, a new imperative for the life sciences:
In the last two decades, the widespread application of genetic and genomic approaches has revealed a bacterial world astonishing in its ubiquity and diversity. It examines how a growing knowledge of the vast range of animal-bacterial interactions, whether in shared ecosystems or intimate symbiosis, is fundamentally altering our understanding of animal biology. It highlights the recent technological and intellectual advances that have changed our thinking about five questions: how have bacteria facilitated the origin and evolution of animals; how do animals and bacteria affect each other’s genomes; how does normal animal development depend on bacterial partners; how is homeostasis maintained between animals and their symbionts; and how can ecological approaches deepen our understanding of the multiple levels of animal-bacterial interaction and to include investigations of the relationships between and among bacteria and their animal partners as we are going to seek a better understanding of the natural world.
Microbiome: Science comes to life:
Farmers have long tried to improve the chemical and physical condition of their soils, seeking to make more nutrients available to their plants, to retain more moisture in the soil, and to ease the growth of plant roots. But they have typically ignored the role of the teeming diversity of fungi and bacteria in the soil. Now, however, soil biologists are beginning to understand the significance of the interactions at work in the microbiome surrounding plants' root systems. Recent research has shown, for example, that major food crops can be made dramatically more stress tolerant by transplanting into them various microbiota, such as fungi or bacteria, that colonize other species.
Harnessing phytomicrobiome signaling for microbiome engineering:
Higher plants have evolved intimate, complex, subtle, and relatively constant relationships with a suite of microbes, the phytomicrobiome. This intercommunication dictates aspects of plant development, architecture, and productivity. Understanding this signaling via biochemical, genomics, proteomics, and metabolomic studies have added valuable knowledge regarding the development of effective, low-cost, eco-friendly crop inputs that reduce fossil fuel intense inputs. This knowledge underpins phytomicrobiome engineering: manipulating the beneficial consortia that manufacture signals/products that improve the ability of the plant-phytomicrobiome community to deal with various soil and climatic conditions, leading to enhanced overall crop plant productivity. The goal of microbiome engineering is to manipulate the microbiome toward a certain type of community that will optimize plant functions of interest. For instance, in crop production, the goal is to reduce disease susceptibility, increase nutrient availability increase abiotic stress tolerance and increase crop yields.
Signaling in the phytomicrobiome:
Virtually every plant part is colonized by microorganisms, including bacteria, archaea, fungi, collectively designated as the plant–microbiome or phytomicrobiome.
Microorganisms are a key component of the plant, often inextricable from their host and the plant–microbiome is thought to function as a meta-organism or holobiont. Over the last few decades, we have learned that plants and microbes can use molecular signals to communicate. This is well-established for the legume-rhizobia nitrogen-fixing symbiosis and reasonably elucidated for mycorrhizal associations. Bacteria within the phytomircobiome communicate among themselves through quorum sensing and other mechanisms. Plants also detect materials produced by potential pathogens and activate pathogen-response systems. This intercommunication dictates aspects of plant development, architecture, and productivity.
Plant Conditioners [Biostimulants]:
Biostimulants may either directly interact with plant signaling cascades or act through stimulation of endophytic and non-endophytic bacteria, yeast, and fungi to produce molecules of benefit to the plant. These are the substances and materials with the exception of nutrients and pesticides, which, when applied to plant, seeds or growing substrates in specific formulations, have the capacity to modify the physiological processes of plants in a way that provides potential benefits to growth, development and/or stress response to enhance the nutrition efficiency and/or stress response.
Arbuscular Mycorrhizal Fungi [AMF] :
AMF support plant nutrition by absorbing and translocating mineral nutrients beyond the depletion zones of plant rhizosphere (biofertilizers) and induce changes in secondary metabolism leading to improved nutraceutical compounds. In addition, AMF interferes with the phytohormone balance of host plants, thereby influencing plant development (bioregulators) and inducing tolerance to soil and environmental stresses (bioprotector). Maximum benefits from AMF activity will be achieved by adopting beneficial farming practices (e.g. reduction of chemical fertilizers and biocides), by inoculating efficient AMF strains and also by the appropriate selection of plant host/fungus combinations.
Biostimulants and Biocontrol strategies :
Biostimulants can be used as a tool to complement the use of chemical inputs, by involving non-living-based products, or living-based products Elicitors and semiochemicals are considered to be the most promising tools for inducing plant resistance to various diseases and enhancing natural predation, respectively. These tools are still difficult to use because of their lack of reliability in the field and their uneasy integration in the cropping systems. Maintaining these performances is supported by research through the development of new tools to increase the tolerance of plants to biotic and abiotic stresses.
Bacterial selection for Biological control of plant disease:
As agricultural production intensified over the past few decades, producers became more and more dependent on agrochemicals as a relatively reliable method of crop protection helping with economic stability of their operations. However, increasing use of chemical inputs causes several negative effects, i.e., development of pathogen resistance to the applied agents and their nontarget environmental impact. Furthermore, the growing cost of pesticides, particularly in less-affluent regions of the world, and consumer demand for pesticide-free food has led to a search for substitutes for these products. Biological control is thus being considered as an alternative or a supplemental way of reducing the use of chemicals in agriculture. There has been a large body of literature describing potential uses of plant-associated bacteria as agents stimulating plant growth and managing soil and plant health, the most widely studied group of PGPB are PGPR colonizing the root surfaces and the closely adhering soil interface, the rhizosphere, some of these PGPR can also enter root interior and establish endophytic populations. Many of them are able to transcend the endodermis barrier, crossing from the root cortex to the vascular system, and subsequently thrive as endophytes in stem, leaves, tubers, and other organs, PGPB, PGPR have provided a greater understanding of the multiple facets of disease suppression by these biocontrol agents
Benefits Of Biostimulants :
"Biostimulants," often used in plural form, is a broad term that literally means a group of ingredients that stimulate life. Stimulate plant responses and work In all weather conditions, Increase microbial root protection from soil pathogens, Increase natural plant toxins, repelling pests, Improve drought tolerance, Stimulate plants' immune system, Detoxify chemical residues and heavy metals, Enhances fertilization and reduces leaching, Produce deeper roots, Improve stress tolerance, Accelerate establishment, Increase soil nutrient reserve, Increase profits, cut operating costs, lead to 50% reduction in fertilizer.
Horticulture hydroponics :
In horticulture, hydroponics is a form of agriculture where plants are not grown in soil, but rather in trays or grow beds fed by a constant flow of nutrient solution. A hydroponic system refers to the tools and equipment that are packaged together in order to grow plants hydroponically. Hydroponics is one the fastest rising sectors in the horticultural industry. It is, however, a contested one.
The globalization of cannabis cultivation- A Growing Challenge:
Contemporary cannabis cultivation takes many different forms with variations in approach identifiable both within and between different countries, a simple typology of modern cannabis cultivation might, therefore, be “old” or “traditional” cultivation, cultivation in the developing world began to take on new dimensions, primarily for domestic consumption. With knowledge and technology (grow-lights, hydroponics, etc.) gradually becoming easily available, opportunities to cultivate cannabis grew. Global patterns of cannabis cultivation have followed a fascinating development, from highly concentrated production in certain developing countries to decentralized production in almost every country around the world. It is with the emergence of modern patterns of cannabis use in the developed world that we have seen major changes in patterns of cannabis production. As demand for cannabis increased globally, fuelled by the developments of the “counter-culture”. Cannabis cultivation may be too easily done, with demand for the product, alongside the knowledge and techniques needed for growing, too widespread to expect anything different.
Joshua Lederberg, who coined the term “Microbiome”, to signify the ecological community of commensal, symbiotic, and pathogenic microorganisms that literally share our body space.
A microbiome is the community of microorganisms such as bacteria, archaea, fungi, as well as viruses that inhabit an ecosystem or organism. Microorganisms dominate all other life everywhere scientists have looked, including the human body, the Earth’s soils and sediments, the oceans and fresh waterways, the atmosphere and even extreme environments such as hydrothermal vents and subglacial lakes. Scientists also use the term microbiome to refer to all these genes associated with those lifeforms.
We, humans, are mostly microbes, over 100 trillion of them. Microbes outnumber our human cells ten to one. The majority live in our gut, particularly in the large intestine. The number of genes in all the microbes in one person’s microbiome is 200 times the number of genes in the human genome. The microbiome may weigh as much as five pounds, Microbiomes have several things in common. Their inhabitants are plentiful and diverse, varying from place to place and even from person to person. They are also dynamic, changing in response to factors such as diet or climate. Finally, they are interconnected with their host, engaged in a give-and-take relationship that is often beneficial, even essential, to both the host and their resident microorganisms.
Both inside and out, our bodies harbor a huge array of micro-organisms. While bacteria are the biggest players, these are dubbed the human microbiota. Your body’s microbiome is all the genes your microbiota contains. The bacteria in the microbiome help digest our food, regulate our immune system, protect against other bacteria that cause disease, and produce vitamins including B vitamins B12, thiamine and riboflavin, and Vitamin K, which is needed for blood coagulation.
Microbiome: The complete genetic content of the microbiota
What is meant by Microbiota:
A microbiota includes bacteria, archaea, protists, fungi, and viruses. A microbiota is an "ecological community of commensal, symbiotic and pathogenic microorganisms" found in and on all multicellular organisms studied to date from plants to animals. The synonymous term microbiome describes either the collective genomes of the microorganisms that reside in an environmental niche or the microorganisms themselves
Aren’t microbes supposed to be dangerous:
Some are pathogens, but others only become harmful if they get in the wrong place or boom in number, and some are very useful to the body – such as by helping to break down the array of sugars found in human breast milk. “These sugars are not broken down by the infant,” other key roles of our microbes include programming the immune system, providing nutrients for our cells and preventing colonization by harmful bacteria and viruses.
Where do microbes come from?
We are sterile when we are in utero, and as we are being born, as we emerge through the birth canal from our mums, we get this handover bacteria. It is like a gulp at birth. Those bacteria are really important for starting the whole process during pregnancy a mother’s microbiome shifts, apparently to an optimum mix for offspring. If you are not born by vaginal delivery but are born by cesarean section, things start off being different
Why microbiome become such a hot topic for research?
As researchers begin to understand what constitutes a healthy microbiome, they are also learning how imbalances in microbial communities can cause disease and how to restore the balance as a means of treatment. There are strong associations between the human gut microbiome and conditions including obesity, type-2 diabetes, cirrhosis, rheumatoid arthritis and inflammatory bowel disease. Researchers have already restored the “normal” human gut microbiome using fecal transplants to cure recurrent infections with the bacterium Clostridium difficile. There are still other associations between the gut microbiome and disorders including anxiety, depression, and autism that researchers are still exploring.
Researchers are just beginning to study the microbiomes in a systematic way. They are trying to understand what makes a healthy microbiome, including what microbes are present and what those microbes are doing, as well as how microbiomes change over time, how such communities stay balanced and how changes to microbiomes impact human or environmental health. Microbiome research is even important for space exploration and planetary science which indulges more interest towards the Microbiome research.
Where do microbiomes exist?
The microbiome varies greatly among
The Human Microbiome:
The microbiome is essential for human development, immunity, and nutrition. It is is the collection of trillions of microbes living in and on the human body. It consists of about a thousand different bacterial species that reside in the mouth, gut, and vagina, and on the skin. These microbes and the genes they express make us a “superorganism” composed of human and microbial cells.
The Earth Microbiome:
The Earth’s soils host microbial communities. Diversity in these underground habitats is tremendously high, with estimates of the soil microbial soil, researchers have found that microbes are essential for supporting plant life, mediating uptake and entry of nutrients into the food chain, cycling carbon, and nitrogen, breaking down pollutants.
The Ocean Microbiome:
Ocean Microbes remove most of the methane from the world’s oceans. These tiny organisms are highly specialized and optimized for their environment, where they provide energy and chemical building blocks to larger life forms. They also produce half of the oxygen we breathe and recycle about the same proportion of carbon dioxide from the atmosphere. The extremely small organisms or “nanoplankton” that perform these essential services form the “nanobiome.”
The Atmospheric Microbiome:
Even the atmosphere has a microbiome. Microbes in the atmosphere are everywhere, vary from place to place and can remain suspended for weeks. The atmosphere transports these microbes, either freely or attached to particles such as dust, around the globe, a phenomenon that may impact agriculture, infectious disease, human health, clouds, precipitation and the water supply.
Researchers mapping the microbiome are discovering previously as an uncharted species and genes. The conviction for a central role of microbiology in the life sciences has been growing exponentially, and microbial symbiosis is advancing from a subdiscipline to a central branch of knowledge in the life sciences.
Travel Guide
Conference Venue Details: https://microbiome.conferenceseries.com/venue-hospitality.php
Find out what you need to visit Canada as a tourist or business person, how to extend your stay in Canada and what documents you need to carry with you to transit through Canada.
Application submission:
Canada does not have a visa office in every country so it is important that Delegates/Attendees visit the website of the visa office responsible for processing their visa applications. Information is available on the website on how to submit a visa application and the documentation required.
Delegates/Attendees are encouraged to submit their visa applications well in advance of the date of the event at a Visa Application Centre or on-line E-applications (e-Apps).
E-Apps
This system allows clients to submit applications online.
Delegates/Attendees that need a visa but require their passport for other travel purposes are strongly encouraged to submit their visa applications online (e-Apps). Delegates/Attendees that choose to apply online will not have to submit their passport until a decision has been taken on their applications. If required, the visa office will send the applicant instructions on how and where to send their passports to finalize the visa process.
Visa Application Centers (VACs):
VACs are commercial service providers authorized by Canada to provide specific services to applicants.
VACs provide a number of services including help applicants fill out forms, answer questions and ensure that applications are complete, thereby reducing unnecessary delays or refusals due to incomplete applications.
Applicants that are required to provide biometrics information as part of the visa application process can do so at a VAC. Additional information on the biometrics requirements is available at the IRCC website.
VACs send applications to Visa Offices and transmit decisions to applicants in a secure and confidential manner. VACs do not process visa applications and play no role in the decision-making process.
Visa Applications Processing Time:
Processing time for visa applications varies depending on the office and the time of the year. Participants should be encouraged to apply early for their visas, and to submit complete applications including all supporting documents.
Please visit the IRCC website for information on the time it takes to process visa applications at the various visa offices.
NEW - Electronic Travel Authorization (eTA)
As of March 15, 2016, visa-exempt foreign nationals are expected to have an Electronic Travel Authorization (eTA) to fly to or transit through Canada. Exceptions include U.S. citizens and travelers with a valid Canadian visa. Canadian citizens, including dual citizens, and Canadian permanent residents are not eligible to apply for an eTA.
However, from March 15, 2016, until fall 2016, travelers who do not have an eTA can board their flight, as long as they have appropriate travel documents, such as a valid passport. During this time, border services officers can let travelers arriving without an eTA into the country, as long as they meet the other requirements to enter Canada. We invite you to consult the IRCC website regularly for information updates on eTA.
Microbiome:
Microbial cells outnumber the human cells in the ratio 10:1 in a health human body. Human microbiome is the study of microbes present in the human body. These are beneficial microbes that carry out functions not performed by any gene in the human DNA and hence, they play a very important role in the human body. For example, these microbes produce certain vitamins that our body cannot make, breakdown our food, equip our immune system to recognize harmful foreign bodies, and produce anti-inflammatory compounds against it.
On the basis of application, the global human microbiome market is segmented into therapeutic and diagnostic. Therapeutic use of human microbiome is increasing with research yielding new applications and usage of human microbiome. The market is thus expected to be inclining towards therapeutic applications in near future. Moreover, microbiome products are considered to be relatively safe, which creates a highly conducive environment for growth of human microbiome industry.
The human microbiome can also be analyzed based on the technology used for conducting research in this field. These technologies include cell culture technology, high-throughput technology, omics technology and computational tools. Products used for research include instruments (analyzers and other laboratory instruments) and consumables (kits and reagents).
Market participants can also understand the human microbiome market dynamics through analysis for different health conditions such as obesity, diabetes, acute diarrhea, mental disorders, autoimmune disorders, cancer, and other diseases.
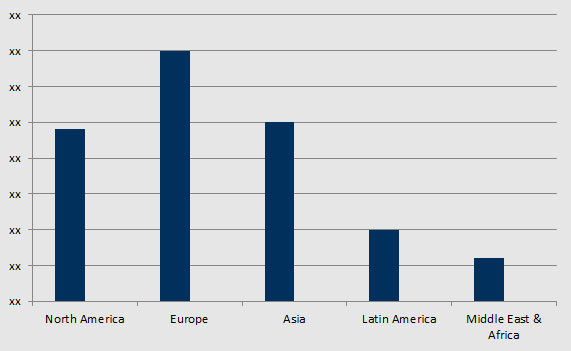
The microbiome market is in growth stage and is rapidly emerging, with promising future. The market is highly developed in North America and Europe, where product and technology is high among the consumers, researchers and healthcare practitioners. Introduction of new products and microbiome modulators in these regions would propel market growth. There is high interest in incorporating microbiome modulators in food and dietary supplements of diet in order to supply the body with the missing dietary supplements. The microbiome technology and products would gain significant traction in the emerging economies of Asia Pacific, Latin America, and Middle East regions. Growing product awareness, rising demand for safe medical products and increasing research activities would be the driving factors for microbiome industry in these regions.
Geographically, the global microbiome sequencing services market is studied for the following regional markets:
North America
U.S.
Canada
Europe
U.K.
Germany
Rest Of Europe
Asia Pacific
China
Japan
Rest of Asia Pacific
Latin America
Brazil
Mexico
Rest of Latin America
Middle East and Africa
GCC
Rest of Middle East and Africa
Along with quantitative information, qualitative information sets and assessment tools are provided in this study for better analysis of the overall market scenario and future prospects. Information such as market drivers, challenges and opportunities assists the readers for understanding the ongoing trends in the global microbiome sequencing services market. Tools such as market positioning of key players and attractive investment proposition provide the readers with insights on the competitive scenario of the global microbiome sequencing services market. This report concludes with company profiles section that highlights major information about the key players engaged in global microbiome sequencing services market
Furthermore, this report concludes with profiling of key market players currently enjoying prominent position in the vascular grafts market. The key players profiled in this report are Baseclear B.V., Clinical-Microbiomics A/S, Diversigen, Inc., Merieux Nutrisciences Corporation, Metabiomics Corp., Microbiome Therapeutics, LLC, Microbiome Insights, Inc., Molzym GmbH & Co. Kg, MR DNA (Molecular Research Lp), Openbiome, Shanghai Realbio Technology Co., Ltd, Rancho Biosciences, Resphera Biosciences, LLC, Ubiome, Inc., Zymo Research Corp. and others.
Biostimulants:
The global biostimulants market is expected to reach USD 3,040.3 million by 2022, supported by a CAGR of 12.7% during the forecast period of 2017 to 2022.
Geographically, the global biostimulants market is segmented into five major regions, namely North America, Europe, Asia-Pacific, Latin America, and the Middle East and Africa. These regions are further analyzed at a country level with the detailed qualitative and quantitative analysis. The geographical analysis provides detailed insights on the key trends of the industry in the listed regions & countries, identifying the demographic & economic impact, agricultural scenario, government initiatives, government & private investments, and regulatory scenario. The geographical analysis section also comments on the key market players shaping the growth of the industry in the respective countries.
The Europe region is the biggest market of Biostimulants and is estimated to grow at a CAGR of 10.03% over the forecast period to reach a total market value of US$936.700 million in 2022. The Biostimulants market in the region is witnessing growth on account activity being undertaken by the manufacturers. Organized manufacturing industry and improved information dissemination among farmers is further contributing to its large-scale usage in farming. Active regulatory bodies such as EBIC and Biostimulants Coalition works in favor of the industry and, enhanced usage of these products and increased demand in the region.
Geographically, European Biostimulants market has been segmented into U.K., Germany, France, Spain, and Italy. Thesecountriesarekey market for biostimulants in the region owing to the expansion of organic farming due to the burgeoning demand for organic products in these countries. Furthermore, regulations and government initiatives are also boosting the demand or biostimulants in the region.
Europe Biostimulants market has been segmented on the basis of active ingredients, crop type, application, and geography
Following an extensive research on biostimulants and an in-depth analysis of market scenario, the report carries out an impact analysis of the key industry drivers, restraints, trends, and opportunities, listed in the market dynamics segment. Growing need to increase crop yield and quality, growing organic farming and demand for organic food, a wide range of benefits of biostimulants in crop production and growing awareness about environmental safety with the use of biostimulants are the key drivers covered in the report which will support the industry growth in the forecast period. However, lack of education and awareness among farmers and various challenges faced by new entrants and small players restricts the growth of biostimulants market. Also, the unavailability of a standardized regulatory framework for biostimulants and scientific & technical difficulties for the development of biostimulants challenges the growth of this market to some extent.
The global biostimulants market is segmented by active ingredients, mode of application, form, and by crop type. On the basis of active ingredients, the market is segmented as acid based, extracts based, microbial amendments, trace minerals and vitamins, and others; On the basis of mode of application, the market is segmented as foliar spray, soil treatment, and seed treatment; and on the basis of formulation, the market is segmented as liquid form and dry form. The report further gives an extensive outlook on the application of biostimulants on various crops. These crop types include row crops, fruits and vegetables, turf and ornamentals, and other crops. The crop type segment further briefly talks about the trends impacting the growth of these markets and the benefits involved with the use of biostimulants on these crops.
Overview of primary interviews
The report also includes the competitive landscape based on an extensive assessment of the key strategic developments adopted by leading market participants in the industry over the past 3 years (2014-2017). The study has also incorporated the detailed profiles of 25 top market players across the value chain of the industry, including detailed financial performance, product offerings, and strategic growth initiatives of each player.
The key players in the global biostimulants market are BASF SE, Valagro SpA, Biolchim S.p.A., Marrone Bio Innovation, Monsanto Company (Part of Bayer), Trade Corporation International ( Part of Sapec Group), Plant Health Care PLC, Syngenta (Subsidiary of ChemChina), Biostadt India Limited, Andermatt Biocontrol AG, BioWorks, Inc., Novozymes, Inc., Koppert B.V., Italpollina S.p.A., Micromix Plant Health Ltd., Arysta Life Science Corporation (Subsidiary of Platform Specialty Products Corporation), FMC Corporation, Bioatlantis Ltd, Omex Agruifluids Limited (Subsidiary of Omex Group), Haifa Chemicals Ltd., Agrinos AS, Camson Bio Technologies Limited, Som Phytopharma India Limited, EuroChem Group AG, and SICIT 2000 SpA
Market size in North America for Biostimulants:
North America Biostimulants market is projected to grow at a CAGR of 9.72% over the forecast period to reach US$716.3 million in 2022. The biostimulants industry is witnessing considerable growth in the North American region on account of remarkable biotechnological growth in the area of enzymes production, and acid extraction from natural products, and thereby enabling the production of efficient biostimulants. Moreover, advanced molecular biology and genetic engineering are also assisting for bioreactor-based amino and other acids production. Furthermore, growth in environment conscious consumers is resulting in increased demand for organic food products, and in turn, is boosting the Biostimulants market in the region.
Finally, the competitive intelligence section deals with major players in the market, their market shares, growth strategies, products, financials, and recent investments among others. Key industry players profiled as part of this section are Isagro S.p.A, Taminco Corp., Valagro S.p.A, Italopolina S.p.A, and Koppert BV among others. Biostimulants market has been segmented on the basis of active ingredients, crop type, application, and geography like Europe.
Conference Highlights
- Human Microbiota
- Microbiota in early life shapes the immune system
- Gut Microbiota
- Modulation of Microbiota
- Bioinformatics for Microbiome
- 16S sequencing & Analysis
- Plant Conditioners [Biostimulants]
- Arbuscular mycorrhizal Fungi [AMF]
- Biostimulants and Biocontrol strategies
- Benefits of Biostimulants
- Horticulture Hydroponics
- Myocardial infarction and gut microbiota: An incidental connection
- Signaling in the phytomicrobiome
- Animals in a bacterial world, a new imperative for the life sciences
- The globalization of cannabis cultivation- A Growing Challenge
- Harnessing phytomicrobiome signaling for microbiome engineering
- Microbiome: Science comes to life
- Bacterial selection for Biological control of plant disease
- Prebiotics and Probiotics
- The Dynamic Microbiome
- Microbiome as a human organ
- The Microbiome as a Therapeutic Target for Metabolic Diseases
- Pharmacogenomics
- The role of the microbiome in cancer development and therapy
- Interplay between diet, gut microbiota, epigenetic events, and colorectal cancer
- Role of microbial communities in the pathogenesis of periodontal diseases and caries (Oral Microbiome)
- The Skin Microbiome
- Skincare products with probiotics worth the hype
- Pediatric Microbiome [Vaginal Delivery Versus C-Section]:
To share your views and research, please click here to register for the Conference.
To Collaborate Scientific Professionals around the World
| Conference Date | October 15-16, 2018 | ||
| Sponsors & Exhibitors |
|
||
| Speaker Opportunity Closed | Day 1 | Day 2 | |
| Poster Opportunity Closed | Click Here to View | ||
Useful Links
Special Issues
All accepted abstracts will be published in respective Our International Journals.
- Journal of Microbial & Biochemical Technology
- Journal of Agricultural Science and Food Research
- Journal of Bioremediation & Biodegradation
Abstracts will be provided with Digital Object Identifier by








































